To put this back into the context of our high school friends, let's say the two atoms meeting and bonding are a vivacious theater student and a more reserved math student.
The negative charge end of the bond is the more attractive atom, because it can draw more negative (-) electrons. When two atoms of the same element meet, neither is stronger than the other, and thus neither one is more attractive than the other regarding electrons, and so neither side exhibits the polar pull, and the bond is considered nonpolar. Just as bigger planets can hold bigger moons in their orbits, so it is with atoms: they pull electrons to the best of their ability, but when they come into the orbit of a better puller (a bigger planet), that atom will pull more electrons into its orbit (and steal the lesser planet's moons). This occurs when the two atoms bonding are from two different elements atoms from the same element do not form polar bonds. These types of bonds are what designates polar molecules from nonpolar ones. Polar bonds are bonds where the electrons are not shared equally between two atoms. Whereas before they had been two separate elements, strangers, now a new substance has formed: a friendship. This sharing means there is a bond of friendship (the covalent bond). Two students (atoms) come close to one another and share their lunch (electrons). To visualize: think of these bonds like the bonds of friendship.
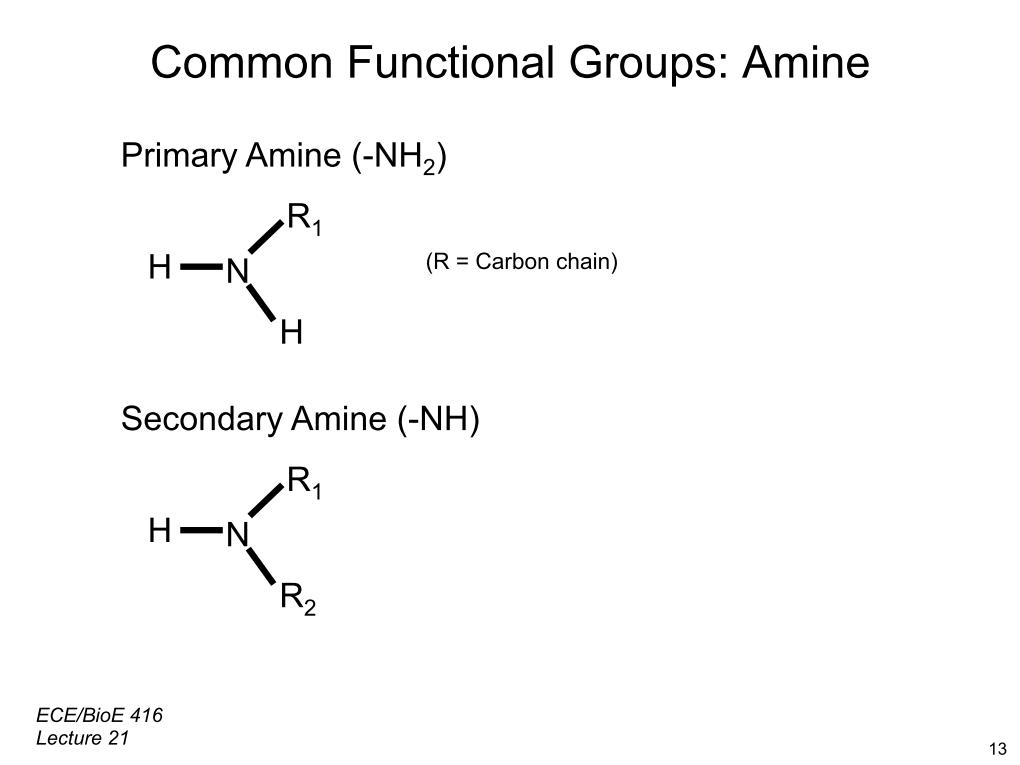
Covalent bonds can exist as polar or nonpolar compounds, and all bonds that are polar or nonpolar in nature are by definition covalent. That creates the connection that then results in a new substance. The bonds form in only one way: when an electron becomes shared by two elements. Covalent BondsĬovalent bonds form the base of the chemical reactions between atoms of different elements. We will start with those definitions for the bond type category-get ready for some high school clique metaphors to help you visualize all this organic chemistry. nonpolar bond types refers to their covalent bonds.
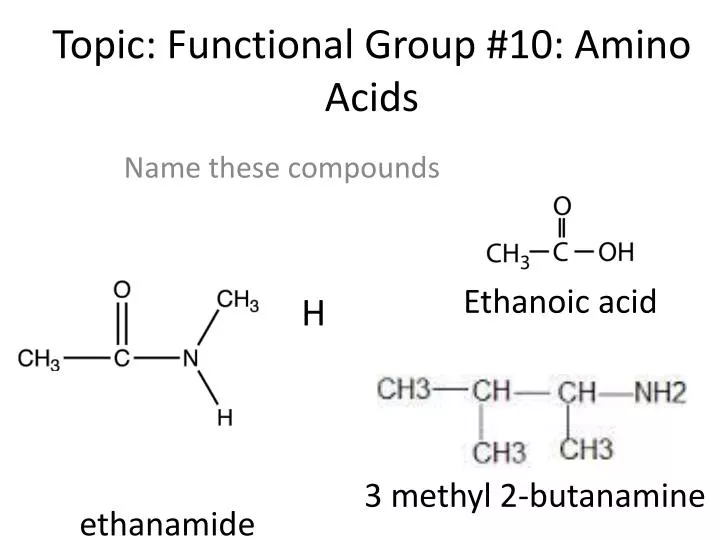
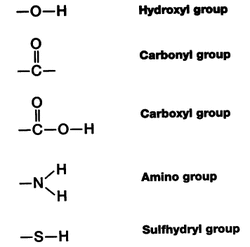
AMINO FUNCTIONAL GROUP PLUS
There are their names, their three-letter codes, their one-letter codes (as used in drawing models of molecular bonds), plus their bond type and properties. The essential aminos are designated with an asterisk (*). Some Basic Reference Definitionsīelow are the names of the 20 common amino acids in the body, nine of which are essential amino acids, meaning humans must eat or otherwise consume them to get what we need to function. This article will help to clarify the structure of amino acids, their functional groups, and what it all has to do with the creation of protein. So you've heard of amino acid functional groups and you want to understand them better.
